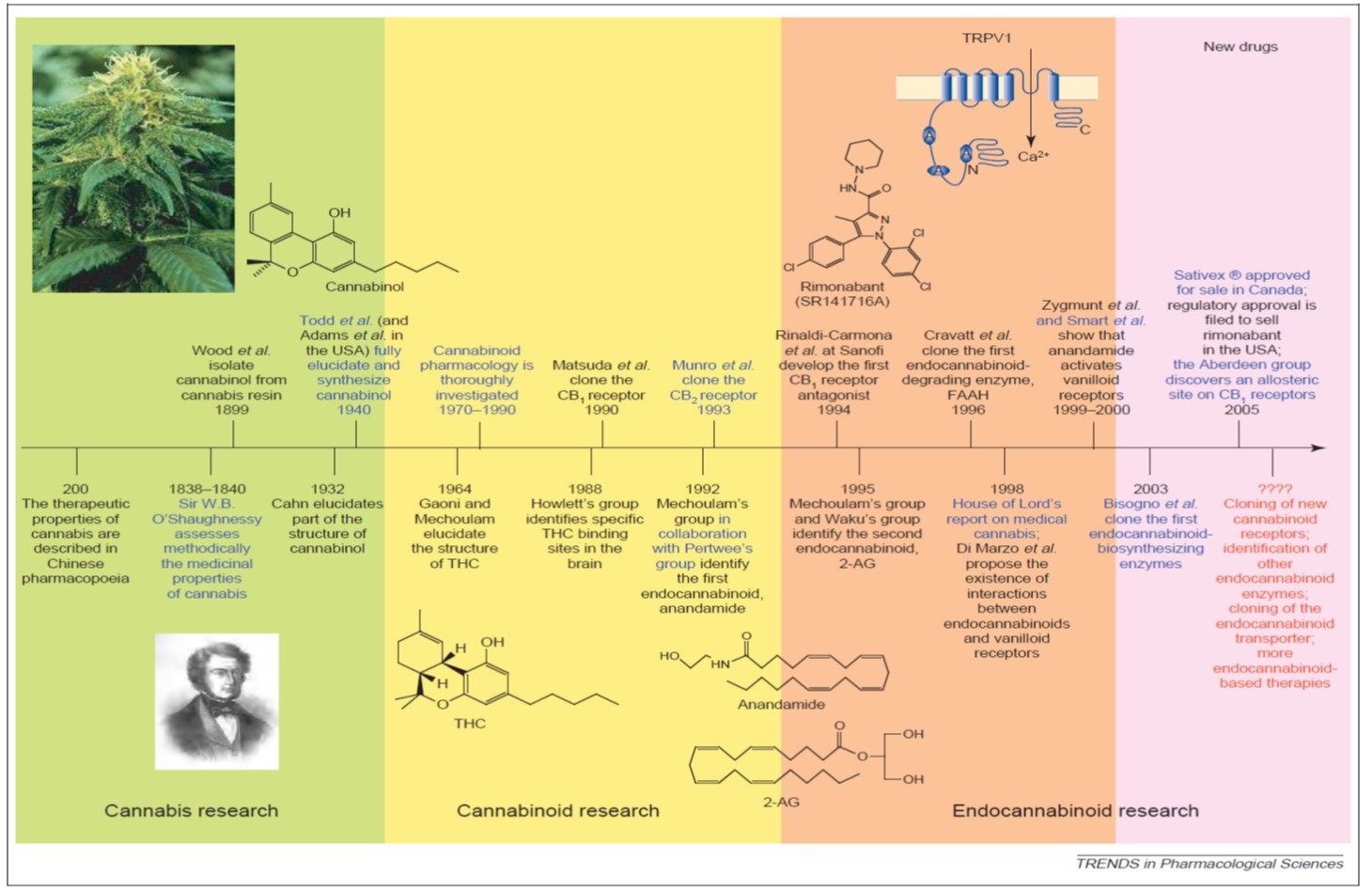
Cannabis sativa is a plant that man has cultivated for both its medicinal as well as its psychoactive properties for many thousands of years. (You may have heard of hemp as well, but that's a much longer story ...) It is indigenous to temperate regions of Asia, so its earliest known use as a medicine traces back to ancient Chinese and Indian civilizations. In the 1800s, "Cannabis indica" (aka Indian hemp, Indian cannabis) was introduced into Western medicine primarily by an Irish physician named William O'Shaughnessy (who visited Calcutta in the 1840s) and was a common ingredient in every physician's kit bag by the early 20th century.
By 1851, it had been added to the US Pharmacopeia (removed by the 1940s) and there were many medical studies and scientific papers published during this time, culminating in the first conference on its use as a medicine held in 1860 by the Ohio State Medical Society. Cannabis fell out of use in medicine by the 1930s for many reasons, not the least of which was the introduction of morphine and the hypodermic needle - both of which became immediately more useful to control pain, for example. Oh, and a little thing called the Marihuana Tax Act of 1937 (read about the history of this).
One can peruse a compressed timeline of key research events above; however, there have been at least two publication surges since the 1800s. The first surge was in the 1960s when THC and cannabidiol (CBD) were discovered and characterized and the second was in the 1990s, when the early discoveries of the endogenous cannabinoid signaling system ("endocannabinoid system," or ECS) were published. Since then, there has been a a slow, steady and sustained increased in annual publications in the field of cannabinoid research from roughly 300 articles per year to over 2,000 per year today (my research, data not shown or published).
One simply cannot underestimate the importance of the ECS to human biology. While there is still much to learn about the system, our current expanded view of this retrograde signaling system is referred to as the "endocannabinoidome," or ECD. It has been found to operate in virtually every tissue and organ system in the body that has been investigated. As one can see below, the ECD is composed of a series of receptors (CB1, CB2, TRPV1 and several GPRs), endogenous signalling molecules (or "endocannabinoids" [eCBs] like anandamide ... there are 8 others as well), membrane transporters and enzymes to break down the eCBs once their job is done.
More about the ECD:
- It has been shown to play key role in “fine tuning” metabolic and homeostatic mechanisms (It makes sure everything runs smoothly from a biochemical point of view).
- It has links to and potential signalling roles in the immune system, digestive system, cardiovascular system, nervous system, skin and skeletal system (for starters!!).
- It plays a key role in chronic disease, including tissue degeneration and remodeling (in bone), inflammation (including in the brain, which serves a neuroprotective role) and in controlling all types of pain.
- It is interconnected with other lipid and non-lipid signaling systems (and other regulatory networks) in a very complex and still not-quite-understood manner.
The cannabinoids (like THC, CBD, etc.) found in Cannabis sativa are referred to as phytocannabinoids (pCBs) and synthetic derivatives (synthetic cannabinoids, sCBs) have been prepared in the laboratory and approved by FDA as drugs. There are essentially three sCBs on the market: Marinol, Cesamet and Sativex. The first two are 100% synthetic THC (in pill form) and the final is a pCB extract, formulated as a sublingual (under the tongue) spray. Another drug currently under investigation is Epidiolex, which is 100% CBD and being examined in clinical trials with children with intractable/severe epilepsy. In fact, many states have now allowed the use of CBD oil to treat pediatric epilepsy that is resistant to conventional treatments. Extracts hold great promise for research due to the "entourage effect," which is the phenomenon described in the literature where more benefits are found from administration of cannabinoids and terpenes together (also found in Cannabis sativa ... about 150 are known to exist and these impart the smell and taste to the plant's flowers and resin) than from THC alone.
Many states have now also instituted state-based programs that regulate the cultivation, processing, distribution and dispensing of medical cannabis products to patients. Patients going through the Veterans Administration health care system; however, generally do not have access to cannabinoid medications due to federal law and policy conflicting with these state-based programs. This is unfortunate as there are many common conditions that our nation's veterans present to VA physicians with, some of which are resistant to conventional treatments. Most states with a medical cannabis regulatory program maintains a list of "treatable conditions" for which cannabinoids may prescribed. (Maryland comes to mind as a notable exception, a state that will not publish such a list and will rely upon judgments made by state-registered physicians engaged in a "bona fide" doctor-patient relationship.)
Some of these conditions include:
- Pain control
- Both human and animal studies show that cannabinoids act as "analgesic" agents and the efficacy of individual products (how well it works) is variable & depends on route of exposure (i.e. oral, inhalation). Analgesic effects have been noted for neuropathic pain, inflammatory pain and cancer pain. Cannabinoids act synergistically with opioids -- currently, the only analgesic for treating severe and chronic pain -- and can act as an “opioid sparing agent” (lower doses, fewer side effects) while having an improved safety profile (i.e. NO OVERDOSES from cannabinoids. Much lower potential for abuse.)
- A systematic review of clinical trial data for treating chronic non-cancer pain from 2011 stated: "Overall the quality of the trials was excellent. 15 of 18 trials that met the inclusion criteria demonstrated a signfiicant analgesic effect of cannabinoid as compared to placebo and several reported significant improvements in sleep. There were no serious adverse effects." [Lynch, M. E., & Campbell, F. (2011). Cannabinoids for treatment of chronic non-cancer pain; a systematic review of randomized trials. British Journal of Clinical Pharmacology, 72(5), 735–744. http://doi.org/10.1111/j.1365-2125.2011.03970.x]
- A systematic review from 2015 for treatng chronic non-malignant neuropathic pain stated: "Cannabis-based medicinal extracts used in different populations of chronic nonmalignant neuropathic pain patients may provide effective analgesia in conditions that are refractory to other treatments." [Boychuk, D. G., Goddard, G., Mauro, G., & Orellana, M. F. (2015). The Effectiveness of Cannabinoids in the Management of Chronic Nonmalignant Neuropathic Pain: A Systematic Review. Journal of Oral & Facial Pain and Headache, 29(1), 7–14. http://doi.org/10.11607/ofph.1274]
- The largest and most recent systematic review (looking at all randomized clinical trial data available for cannabinoids) from 2015 stated: "Compared with placebo, cannabinoids were associated with a greater average number of patients showing a reduction in pain" from 8 trials, which the authors characterized as "moderate-quality evidence." [ Whiting, P. F., Wolff, R. F., Deshpande, S., Di Nisio, M., Duffy, S., Hernandez, A. V., … Kleijnen, J. (2015). Cannabinoids for Medical Use: A Systematic Review and Meta-analysis. JAMA, 313(24), 2456. http://doi.org/10.1001/jama.2015.6358]
- Inflammation control
- All classes of cannabinoids show anti-inflammatory activity and the role of THC and the endocannabinoids in this response is well-studied. CBD has been shown to reduce joint, instestinal and neuroinflammation in animal models (including reduction of glucose-induced inflammation in diabetic mice). At least one terpene (caryophyllene) has been found to have anti-inflammatory activity and can bind to one of the ECD receptors. There's even some evidence in acute brain injury models that cannabinoids serve a neuroprotective role by reducing swelling.
- Post-Traumatic Stress Disorder (PTSD)
- PTSD patients have been found to express more CB1 receptors in their brain and thus, the stimulation that cannabis provides at this receptor has been widely found to provide effective short-term relief of symptoms. What is less well known are the long-term impacts as well as the impacts of self-medication and not adhering to prescribed doses. One small clinical trial of 10 patients in 2014 found that cannabis was well-tolerated and reduced symptoms of hyperarousal; however additional clinical trials are ongoing and/or planned. The VA system has reported in a series of publications over many years that, among veterans (seen in VA health care facilities) with co-occurring PTSD and substance use disorders (SUD), cannabis use disorder (CUD) is most diagnosed SUD since 2009. So there appears to be a fine line between controlling symptoms in the short-term and CUD in the long-term. In the meanwhile, CBD is also now being explored as a way to control social anxiety.
- A 2015 study from the VA reported that: "There is convincing evidence from multiple studies for reduced endocannabinoid availability in PTSD." (indicating endocannabinoid-targeted therapies should be explored) [See "Translational evidence for a role of endocannabinoids in the etiology and treatment of posttraumatic stress disorder."]
- Addiction
- This is one of the newer areas of research and both THC and CBD have been investigated for use in treating addiction. Studies done in the last decade have found cannabis appears to reduce cravings for several highly addictive substances, like crack cocaine, opiates and alcohol. Users claim less withdrawal symptoms, fewer side effects (than from conventional addiction treatment substances, like buprenorphine or methadone) and better symptom management. A systematic review of available evidence in 2015 found that some human studies have demonstrated benefit from CBD in treating cannabis and tobacco dependence. Sativex (a blend of equal amounts of THC and CBD) was even found to attenuate cannabis withdrawal symptoms in a 2014 clinical trial and ongoing trials in the US and Canada are investigating use of CBD in treating cocaine, opiod and cannabis addiciton. One very interesting case report from 2015 found a Canadian medical cannabis patient was able to fairly quickly taper down from 30-40 mg of opioids a day (prescribed for post-operative pain following a liver transplant) to 6-8 mg/day by using medical cannabis to control his chronic and severe pain as well as nausea. For pain, a strain with 17% CBD and less than 1% THC was used and for nausea, a strain with 12% CBD and 9% THC was used. [Meng, H., Hanlon, J. G., Katznelson, R., Ghanekar, A., McGilvray, I., & Clarke, H. (2015). The prescription of medical cannabis by a transitional pain service to wean a patient with complex pain from opioid use following liver transplantation: a case report. Canadian Journal of Anesthesia/Journal Canadien D’anesthésie, (January 2016). http://doi.org/10.1007/s12630-015-0525-6]
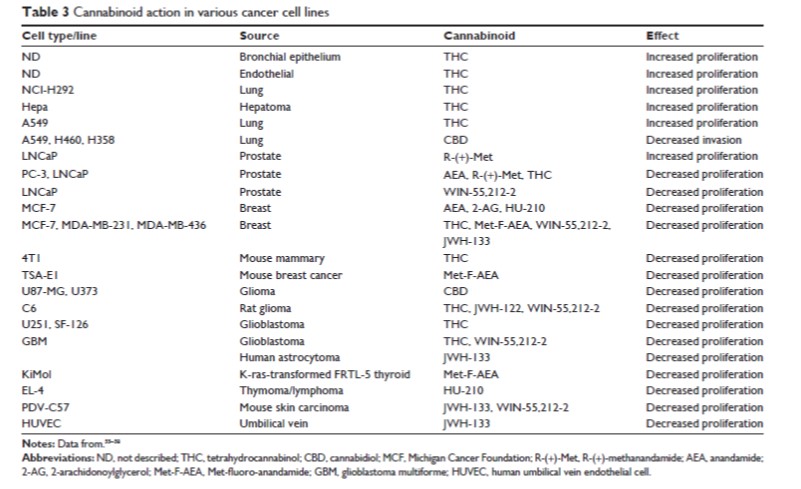
- Cancer
- The first study to be published that showed anti-tumorigenic properties of cannabinoids was in 1975! Since then, palliative care applications have long been recognzied and cannabinoids have played an important role in controlling pain, nausea, vomiting and appetite in cancer patients. Both in vitro and in vivo evidence support the hypothesis that endogenous cannabinoids can reduce proliferation and invasivity of prostate cancer cells; however, there has been much research in a wide variety of cell types and there have been mixed results. Some results can be seen below ... it is worth pointing out that THC itself has been to have both pro-profliferative and anti-proliferative properties.
While the results presented above are promising, they represent but a sliver of the available evidence. It must be stated strongly that additional mechanistic work is needed in animal, in vitro and human systems to more fully understand the role the ECS plays in maintaining "wellness." Even more important is the need for federal funding to run large-scale clinical trials in groups of human patients to understand more fully all the safety and efficacy issues for cannabinoid-based therapies. At this point, large cannabis industry companies like Tilray are funding larger-scale trials. The key to restoring FDA's ability to conduct large-scale trials is the removal of cannabis from the Controlled Substance Act scheduled list of illegal substances with no medical value ... a move that should happened a long time ago.
Essential Reading (all are free downloads):
Modulating the endocannabinoid system in human health and disease: successes and failures (http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3684164/)
Re-branding cannabis: the next generation of chronic pain medicine? (http://www.futuremedicine.com/doi/pdf/10.2217/pmt.14.49)
Cannabidiol as an Intervention for Addictive Behaviors: A Systematic Review of the Evidence. (http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4444130/)
Cannabis - the Israeli perspective. (http://www.ncbi.nlm.nih.gov/pubmed/26426888)
Taming THC: potential cannabis synergy and phytocannabinoid-terpenoid entourage effects. (http://www.ncbi.nlm.nih.gov/pubmed/21749363)
Are cannabidiol and Δ(9) -tetrahydrocannabivarin negative modulators of the endocannabinoid system? A systematic review. (http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4301686/)
Targeting the endocannabinoid system with cannabinoid receptor agonists: pharmacological strategies and therapeutic possibilities. (http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3481523/)
Cannabidiol: pharmacology and potential therapeutic role in epilepsy and other neuropsychiatric disorders. (http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4707667/)
Care and feeding of the endocannabinoid system: a systematic review of potential clinical interventions that upregulate the endocannabinoid system. (http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3951193/)